Summary:
- Merck is an American biopharma powerhouse that has generated consistent compounding growth, increasing its top line and EPS.
- Merck expects to deploy capital through strategic small bolt-on acquisitions, which we are a big fan of, rather than rampant big indiscriminate acquisitions.
- Merck’s late-stage candidate Sotatercept and MK-0616’s clinical data are expected to be presented during March’s ACC conference; we expect the results to be positive.
- We initiate Merck with a Buy rating.

Solskin
Company background
Merck & Co (NYSE:MRK) is an American big pharmaceutical company also known as MSD, outside of the United States and Canada. Merck is a global pharmaceutical company that develops, manufactures, and sells a wide range of healthcare products. The company was founded in 1891 and is headquartered in Kenilworth, New Jersey. The key drivers of Merck’s growth are their strong research and development capabilities, their focus on innovative products, and their established presence in various global markets. Merck’s portfolio includes products in a variety of therapeutic areas, such as oncology, vaccines, infectious diseases, and animal health.
Merck’s key revenue drivers are fairly dependent on Keytruda and their vaccine business:
- Keytruda – is an immunotherapy drug used to treat various types of cancer, including lung cancer, melanoma, and bladder cancer.
- Gardasil – a vaccine that helps prevent certain types of human papillomavirus (HPV), which can lead to cervical cancer and other diseases.
- Januvia – a drug used to treat type 2 diabetes.
- ProQuad – a vaccine that helps protect against measles, mumps, rubella, and varicella (chickenpox).
Merck has been a solid compounder
As shown below, during 2022, Merck’s revenue has grown 22% YOY, and EPS has grown by more than $2, from $5.37 to $7.48. We see this growth trajectory continuing.
Merck IR presentation (Merck IR presentation)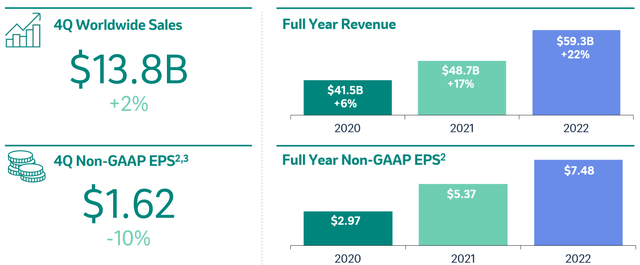
During the Q4 presentation, the company emphasized four key areas for success moving into the next decade, a) enhancing durable growth through focusing on current key growth drivers such as Gardasil and Keytruda, where the company expects ~$11Bn of revenue by 2030; b) small bolt-on business development acquisitions to add value to the current pipeline, c) further expanding leadership in oncology through focusing on Keytruda, d) expediting R&D development in clinical assets (sotatercept, V116 in pneumococcal disease, MRA-4157/V940 in melanoma) with a target of reaching >$10Bn in sales by mid-2030.
Key clinical catalysts expected during the American College of Cardiology conference in March 2023
We believe the American College of Cardiology (ACC) 2023 conference, which is expected to be held around March 4-6, to be a high-impact catalyst for Merck that can move the stock price meaningfully, 5-10%. Many investors were cautious about Merck’s future growth potential based on the company’s concentrated portfolio with a high concentration on Keytruda and Gardasil. We believe the phase 3 STELLAR trial of Sotatercept and phase 2 data from oral PCSK9 will address the aforementioned concerns. We remain confident about the two readouts due to the following reasons.
Sotatercept
Sotatercept is an investigational drug being studied for the treatment of pulmonary arterial hypertension (PAH). PAH is a progressive and life-threatening disease that causes high blood pressure in the arteries that supply blood to the lungs. Sotatercept works by inhibiting a protein called transforming growth factor-beta (TGF-β), which plays a role in the regulation of blood vessel growth and remodeling. By inhibiting TGF-β, Sotatercept may help to prevent the thickening and narrowing of blood vessels in the lungs that occurs in PAH. Last year, Merck announced positive results of the phase 3 STELLAR trial, where the drug has met stat sig improvement in 6MWD, which is a key endpoint for FDA and EMA approval. The degree of improvement would be important for its market positioning; we expect around ~20-25 meter improvement in 6MWD, which we believe should be adequate enough for sotatercept to be positioned as a fourth-line add-on agent to conventional triplet therapy, and we expect it to go up the line of therapy with more RWE data down the line. As PAH is a >$8Bn market where many patients become refractory to existing therapies and rotate around into multiple different combinations, we see a potential peak sales of >$1Bn for a novel drug like sotatercept that can be used as an add-on to the currently available treatments.
Available data on phase 3 STELLAR:
… announced positive top-line results from the pivotal Phase 3 STELLAR trial evaluating the safety and efficacy of sotatercept, an investigational activin receptor type IIA-Fc (ActRIIA-Fc) fusion protein being evaluated as an add-on to stable background therapy for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1). The trial met its primary efficacy outcome measure, demonstrating a statistically significant and clinically meaningful improvement in 6-minute walk distance (6MWD, which measures how far patients can walk in 6 minutes) from baseline at 24 weeks. Eight of nine secondary efficacy outcome measures achieved statistical significance, including the outcome measure of proportion of participants achieving multicomponent improvement (defined as improvement in 6MWD, improvement in N-terminal pro-B-type natriuretic peptide (NT-proBNP) level, and either improvement in WHO FC or maintenance of WHO FC II), and the outcome measure of time to death or the first occurrence of a clinical worsening event (TTCW). The Cognitive/Emotional Impacts domain score of PAH-SYMPACT®, which was assessed as the ninth and final secondary outcome measure, did not achieve statistical significance. The overall safety profile of sotatercept in STELLAR was in general consistent with what has been observed in Phase 2. Results from the study will be presented at an upcoming scientific congress.
We are positive on MK-0616
MK-0616 is an investigational drug being developed by Merck & Co. for the treatment of atherosclerotic cardiovascular disease (ASCVD). ASCVD is a disease that occurs when the arteries become narrowed or blocked due to a build-up of plaque, which can lead to serious conditions such as heart attack and stroke.
The two early-stage phase 1 clinical trials (healthy volunteer study and hypercholesterolemia patients with ~40-60 patients each) showed that MK-0616 was i) safe and well tolerated without any serious adverse events, ii) blood levels of free proprotein convertase subtilisin/kexin type 9 were reduced by 90% from baseline when administered to healthy men, and iii) LDL-c was reduced ~65% from baseline within 14 days, which we believe is very strong, comparable to already approved parenteral proprotein convertase subtilisin/kexin type 9 antibodies (Praluent and Repatha) and siRNA like Leqvio.
Previous data on MK-0616:
A first randomised, double-blind, placebo-controlled, in-human trial assessed the safety and tolerability of single doses of MK-0616 ranging from 10 mg to 300 mg in 60 male participants (aged 18–50 years). MK-0616 was generally safe and well tolerated in this population. No serious adverse events (‘AES’) were reported and only 1 treatment-related discontinuation was observed, a case of maculopapular rash. Drug-related AEs were mostly abdominal discomfort, diarrhoea, dyspepsia, and headache. Free Proprotein convertase subtilisin/kexin type 9 was reduced by more than 80%, regardless of the administered dose of MK-0616. This effect lasted for approximately 24 hours. Free PCSK9 levels returned to baseline levels in 96 hours. In addition, the authors observed that a permeation enhancer (i.e. sodium caprate) improved the absorption of MK-0616 and noted a negative pre-dose food effect.
A second double-blind, placebo-controlled, phase 1 trial evaluated the LDL-cholesterol lowering capacities of MK-0616 in 40 men and women (aged 18–65 years) treated with statin therapy. Patients were randomised 3:1 to 1 of 3 dosing regimens of MK-0616 or placebo (see Figure). The 14-day result displayed no serious AEs, deaths, or discontinuations. Treatment-related AEs were similar to the reported AEs in the first trial, demonstrating a favourable safety profile of the agent. LDL cholesterol was reduced by a maximum of 65% in all experimental conditions, except for the pre-dose food condition (see Figure). This result suggests that low-dose MK-0616 plus low-dose sodium caprate can achieve a major reduction in LDL cholesterol. Larger clinical trials need to confirm the safety and efficacy of MK-0616 in a diverse population.
PCSK9 reduction (Company source) MK-0616 dose response (MK-0616 dose response)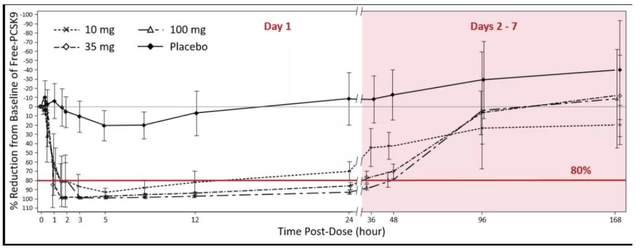
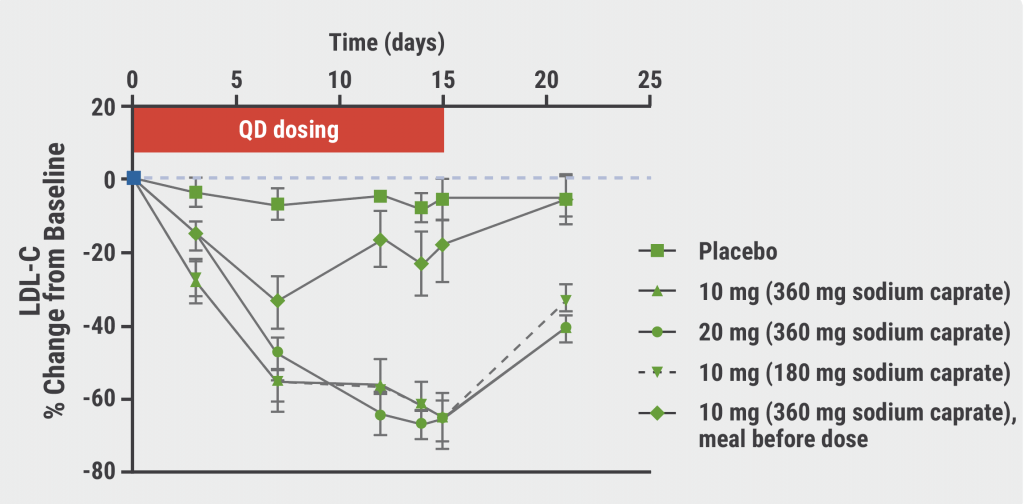
Keeping the previous LDL-c reduction shown in phase 1 in mind, we expect a ballpark of ~50-60% LDL-c reduction in the full phase 2 readout. However, we emphasize that the current data available is fairly limited (only two phase 1 trials), and larger and longer-term studies would be necessary for us to build comfort on the platform.
A quick summary of the ongoing phase 2 trial design of MK-0616 expected to be published in March 2023:
The phase 2 trial was a randomized, double-blind, placebo-controlled study that aimed to evaluate the safety and efficacy of MK-0616 in patients with hypercholesterolemia who were already taking statins. The trial enrolled 423 patients from multiple centers in the United States and Europe. The trial was designed to assess whether MK-0616 could provide additional LDL cholesterol-lowering benefits to patients already taking statins. This is an important question because many patients with hypercholesterolemia do not achieve their LDL cholesterol goals with statin therapy alone. If MK-0616 is found to be safe and effective in this patient population, it could provide a valuable new treatment option for patients with ASCVD.
The patients were randomized to receive either MK-0616 or a placebo for 12 weeks. MK-0616 was administered orally, once daily, at doses of 5 mg, 10 mg, or 20 mg. The primary endpoint of the trial was the percentage change in LDL cholesterol levels from baseline to week 12. In addition to assessing the efficacy of MK-0616, the trial also evaluated its safety and tolerability. Adverse events were monitored throughout the study, and clinical laboratory tests were performed to assess potential changes in liver function, kidney function, and other parameters.
Risks
-
Patent expiration: Like many pharmaceutical companies, Merck and Co faces the risk of patent expiration on key drugs in its product pipeline. Once patents expire, generic competitors can enter the market and erode sales of the branded drug.
-
Regulatory risks: The pharmaceutical industry is heavily regulated, and companies like Merck and Co must navigate a complex regulatory environment to bring new drugs to market. There is always the risk that regulatory agencies may not approve a drug or that the approval process will be delayed.
-
Clinical trial results: Merck and Co invest heavily in research and development to bring new drugs to market, but there is always the risk that clinical trials will fail to demonstrate efficacy or safety. This can lead to costly delays and setbacks in the drug development process.
-
Competition: The pharmaceutical industry is highly competitive, with many companies vying for market share in the same therapeutic areas. Merck and Co face competition from both established players and emerging biotech companies with innovative new drugs.
-
Economic conditions: The pharmaceutical industry is not immune to economic downturns, and Merck and Co’s financial performance may be impacted by macroeconomic factors such as recessions or changes in healthcare policy.
-
Litigation risks: Like many large corporations, Merck and Co face the risk of lawsuits and legal challenges related to its products, patents, or business practices. These legal challenges can be costly and may impact the company’s reputation.
-
Dependence on key products: Merck and Co generate a significant portion of its revenue from a few key products, such as Keytruda. If these products face challenges such as patent expiration, competition, or regulatory hurdles, it could have a significant impact on the company’s financial performance.
Conclusion
We are buyers of Merck based on the company’s continued growth in both top line and EPS, strong global presence and track record in drug commercialization, positive strategic bolt-on acquisitions expected during the next decade replenishing their pipeline, and several positive clinical catalysts (sotatercept and MK-0616) expected in 2023 March.
Disclosure: I/we have no stock, option or similar derivative position in any of the companies mentioned, and no plans to initiate any such positions within the next 72 hours. I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.
Additional disclosure: Biotechvalley Insights (BTVI) is not a registered investment advisor, and articles are not targeted toward retail investors. The content is for informational purposes only; you should not construe any such information or other material as legal, tax, investment, financial, or other advice. Nothing contained in our articles or comments constitutes a solicitation, recommendation, endorsement, or offer by Biotechvalley Insights or any third-party service provider to buy or sell any securities or other financial instruments in this or in any other jurisdiction in which such solicitation or offer would be unlawful under the securities laws of such jurisdiction.
The research and reports made available by BTVI reflect and express the opinion of the applicable BTVI entity as of the time of the report only. Reports are based on generally-available information, field research, inferences, and deductions through the applicable due diligence and analytical process. BTVI may use resources from brokerage reports, corporate IR, and KOL/expert interviews that may have a conflict of interest with the company/assets that BTVI covers. To the best of the applicable BTVI’s ability and belief, all information contained herein is accurate and reliable, is not material non-public information, and has been obtained from public sources that the applicable BTVI entity believes to be accurate and reliable. However, such information is presented “as is” without warranty of any kind, whether express or implied. With respect to their respective research reports, BTVI makes no representation, express or implied, as to the accuracy, timeliness, or completeness of any such information or with regard to the results to be obtained from its use. Further, any analysis/comment contains a very large measure of analysis and opinion. All expressions of opinion are subject to change without notice, and BTVI does not undertake to update or supplement any reports or any of the information, analysis, and opinion contained in them.
